International Consortiums
- PedCon Consortium: Machine Learning models of Bone Growth and morphology of the pediatric knee structures for optimal management of pediatric knee joint disorders.
Summary:
Musculoskeletal knee injury and subsequent surgical treatment is increasing in pediatric and adolescent populations.[1-3] Children who incur these injuries, particularly patellofemoral dislocation and anterior cruciate ligament (ACL) tears, are vulnerable to early-onset osteoarthritis and chronic musculoskeletal impairment through their lifetime.[4, 5] Improved morphological and biomechanical information of the pediatric knee would enhance the evaluation, diagnosis, and treatment of pediatric sport injury and musculoskeletal disease. Additionally, models that predict the progression of bone growth would be invaluable to practitioners making surgical and rehab decisions pertaining to the timing and the method of treatment. For example, the rate of ACL reconstructions is increasing at a significantly higher rate in adolescent populations than in adults.[6] Management of ACL injury in skeletally immature patients is challenging due to continued growth at the distal femoral and proximal tibial physes. Surgery would risk physeal damage and growth cessation whereas delaying surgery until after skeletal maturity may increase the occurrence of chronic instability, chondral and meniscal damage, and decreased function. As such, current management of ACL injuries including operative timing, surgical method and rehabilitation, remain controversial. The goal of restoring knee stability while minimizing the risk of growth disturbance would be greatly enhanced with precision information regarding current and longitudinal physeal growth and fusion timing for individual patients. The number of pediatric patellar instability surgeries is also increasing[3] and similar to pediatric ACL reconstruction, information on bone growth would be vital to patient care and management.[4] Morphological knee information would be especially valuable if it included anatomical risk factors and patient-specific biomechanical implications for ACL loading and injury and patellofemoral instability.
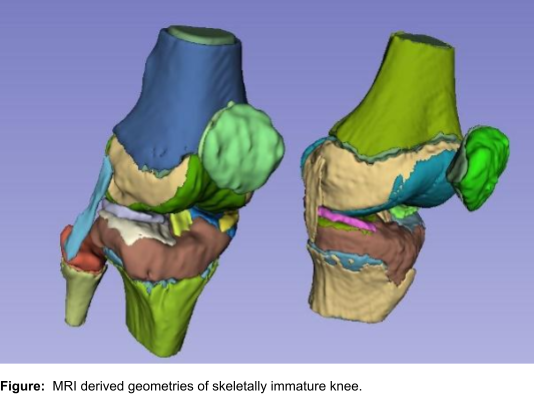
Resolving the translational complexity of the management of pediatric knee disorders would need a confluence of current advances in machine learning, medical image analysis, computational modeling, human movement science, and skeletal growth domains. To this extent, we have formed an international consortium of leading researchers from three institutions (University of Missouri System, USA; University of Cape Town, South Africa; Symbiosis Centre for Medical Image Analysis, Symbiosis International University, India). Researchers involved in this network have complementary knowledge spanning domains such as skeletal growth sciences, machine learning, artificial intelligence, medical image analysis, data-driven statistical modeling, multi-body and finite-element modeling, clinical biomechanics, motion analysis, clinical sciences and pediatric orthopedics. Our research consortium proposes to use a large pediatric medical imaging dataset (magnetic resonance imaging (MRI)) to characterize the morphology, growth, epiphyseal fusion, and biomechanical evaluation of pediatric and adolescent knees. MRI data will be used to create fast, automated, and novel methods for morphometric analysis, skeletal growth prediction, and biomechanical simulation of the developing knee. This would, in turn, provide diagnostic information for the improved management of multiple musculoskeletal injuries and disorders in children.
Aims:
- To characterize three-dimensional morphology of the growth plates and associated structures using image-based bone growth and fusion analysis in pediatric populations:
- To build data-driven models specific to growth and maturation at the knee:
- To identify ACL and PFL growth morphology and relate to biomechanics for management of ACL injury and patellofemoral instability:
References:
- Fabricant, P.D. and M.S. Kocher, Anterior Cruciate Ligament Injuries in Children and Adolescents. Orthop Clin North Am, 2016. 47(4): p. 777-88.
- Nogaro, M.C., et al., Paediatric and adolescent anterior cruciate ligament reconstruction surgery. Bone Joint J, 2020. 102-B(2): p. 239-245.
- Arshi, A., et al., Operative Management of Patellar Instability in the United States: An Evaluation of National Practice Patterns, Surgical Trends, and Complications. Orthop J Sports Med, 2016. 4(8): p. 2325967116662873.
- Clark, D., et al., Adolescent patellar instability: current concepts review. Bone Joint J, 2017. 99-B(2): p. 159-170.
- Mansson, O., et al., Long-term clinical and radiographic results after delayed anterior cruciate ligament reconstruction in adolescents. Am J Sports Med, 2015. 43(1): p. 138-45.
- Dodwell, E.R., et al., 20 years of pediatric anterior cruciate ligament reconstruction in New York State. Am J Sports Med, 2014. 42(3): p. 675-80.